Farmeconomia. Health economics and therapeutic pathways 2026; 27(1): 13-20
https://doi.org/10.7175/fe.v27i1.1604
Original research
Economic Implication of Targeting Higher Trough Level with Extended Half-Life FVIII Prophylaxis
Carla Fornari 1, Stefania Antonacci 2, Stefano Stoppa 3, Lorenzo Giovanni Mantovani 1,4, Giancarlo Castaman 5, Paolo Angelo Cortesi 1,4
1 Research Centre on Public Health (CESP), University of Milano-Bicocca, Monza, Italy
2 U.O.C. Farmacia territoriale, ASL Bari, Bari, Italy
3 Farmacia Ospedaliera, ASST ovest milanese, Legnano, Italy
4 Laboratory of Public Health, IRCCS Istituto Auxologico Italiano, Milan, Italy
5 Center for Bleeding Disorders and Coagulation Careggi University Hospital, Florence, Italy
Abstract
AIM: This analysis aimed to assess the annual International Unit (IU) consumption and costs of prophylaxis therapies with extended half-life recombinant FVIII (EHL-rFVIII), targeting a traditional trough level of 1% and the higher levels of 3% and 5% recommended by recent guidelines and expert consensus, using a theoretical pharmacokinetic model.
METHODS: A pharmacokinetic model was developed to calculate annual treatment IU consumption and costs of EHL-rFVIII products to sustain different trough levels (1%, 3% and 5%) in adult patients with hemophilia A in Italy. The model assumed a one-year perspective and assessed two scenarios: one with lower frequency of administration and one with higher frequency.
RESULTS: The related annual treatment per-patient cost increased as the trough level increased. The per-patient annual cost was €117,811 to achieve a trough level of 1%, €351,543 to achieve a 3% trough level and €585,905 to achieve a 5% trough level. The increase in dose frequency (alternative scenario), that is the reduction of time between infusions, reduced the estimated doses to achieve the pre-defined FVIII levels.
CONCLUSIONS: These results suggest the need for further discussion about the trough levels to target with the actual EHL-rFVIII and the prophylaxis dose and regimen selection for each patient, also considering all treatment options available and the economic implications.
Keywords
Hemophilia A; Prophylaxis; rFVIII; Pharmacokinetic; Health economics
Carla Fornari
Received 18 December 2025
Accepted 13 February 2026
Published 12 March 2026
Introduction
Hemophilia A is an inherited bleeding disorder caused by variable deficiency in clotting factor VIII (FVIII) [1]. In severe cases (FVIII < 1%), spontaneous or injury-related bleeding can occur, primarily affecting joints but also other tissues. Repeated and prolonged spontaneous bleeding episodes experienced by hemophilia A patients can lead to significant morbidity, including disabling musculoskeletal damage and hemophilic arthropathy [2,3].
Historically, the primary treatment for hemophilia A has been replacement therapy, involving intravenous administration of standard half-life recombinant FVIII products (SHL-rFVIII) and plasma-derived FVIII (pdFVIII) [4]. Early initiation of prophylaxis, involving regular infusions of clotting factors to prevent bleeding episodes, has emerged as the standard of care, since it significantly reduces joint damage and the risk of life-threatening bleeds [2,3].
While effective, this approach often requires frequent injections, which can be burdensome and costly. Furthermore, the pharmacokinetics (PK) characteristics of SHL-rFVIII and pdFVIII products and the associated treatment burden (number of intravenous administrations per week) have limited the possibility of achieving trough plasma levels higher than 1-2% and have raised issues of treatment adherence [5-7]. Recent research has led to a shift towards personalized prophylaxis regimens, tailored to individual patient needs. This approach aims to improve treatment outcomes while minimizing the burden on patients and has been supported by the introduction of new treatment options, including extended half-life factor concentrates and non-replacement factors.
The introduction of extended half-life recombinant FVIII products (EHL-rFVIII) provides new prophylaxis strategies to improve convenience and adherence through prolongation of the interval between infusions or by increasing trough levels to enhance bleed prevention while maintaining current time intervals [5-7]. Recent guidelines and expert consensus indicate the opportunity to achieve a minimum trough level of 3-5% with the available EHL-rFVIII products to improve the prevention of bleeding and preserve joint function [4,7]. These recommendations are also supported by recent clinical trial results that show how increasing FVIII levels can reduce the bleeding rate and increase the proportion of patients with zero bleeds [8]. However, these new thresholds require a higher consumption of IU of EHL-rFVIII and/or an increase in administration frequency, with a possible impact on treatment costs. The dosage and frequency of infusion to be used for each individual EHL-rFVIII product depends on both the threshold of FVIII concentration that is desired to be reached in patients, and the pharmacokinetics of the individual products.
The objective of this study is to estimate the annual International Unit (IU) consumption and cost of prophylaxis therapies with EHL-rFVIII targeting the traditional trough level of 1% compared to the new strategies that aim to achieve a 3% or 5% trough level, using a theoretical pharmacokinetic model.
Methods
A PK computation model was developed in Microsoft Excel to calculate annual treatment cost of EHL-rFVIII products to sustain a specified trough level in patients with hemophilia A in Italy. The model operates on two levels: the first one computes the treatment dose needed to achieve a pre-specified trough level at a defined infusion frequency, while the second level computes and compares the annual infusion units (IUs) per-patient and costs of different setups defined by trough levels. The model focuses on adult hemophilic patients and assumes a one-year time horizon. The EHL-rFVIII products analyzed are turoctocog alfa pegol, rurioctocog alfa pegol, efmoroctocog alfa, and damoctocog alfa pegol.
Dose definition (first level)
The model applies a common individual PK equation to compute the needed dose to achieve three pre-specified trough levels of 1%, 3%, and 5% at a defined treatment dose frequency [9]. In the equation (in Supplementary Figure S1) the dose computation depends on PK individual parameters, as clearance (Cl) and volume of distribution at steady state (Vss), dosing interval (ῖ) and time since dose (t). For the purpose of this study, t was assumed to be equal to the dosing interval. The dose frequency for each product was derived from the summary of product characteristics (SmPC) and, for efmoroctocog alfa, from a phase 3 clinical trial [10]. The dose frequencies imputed into the model were: every four days for turoctocog alfa pegol [11], twice a week for rurioctocog alfa pegol [12], 1.9 infusions per week for efmoroctocog alfa [10], and every 5 days for damoctocog alfa pegol [13].
Cl and Vss are strictly related to individual characteristics, and they quantify the drug processing capacity and the distribution of the drug within the human body, respectively. In our model, PK parameters were populated using data from population PK studies comparing EHL-rFVIII products: specifically, one comparing damoctocog alfa pegol and efmoroctocog alfa [14] and the another one comparing damoctocog alfa pegol and rurioctocog alfa pegol [15]. As mean PK parameters vary among studies, due to high individual variability of parameters and different characteristics of the analyzed samples, we recalibrated PK parameters to ensure estimates for EHL-rFVIII products were based on comparable populations. The study by Solms and colleagues (2020) [15], which reported slower pharmacokinetic for damoctocog alfa pegol, was used as the main reference for recalibration. No direct comparison of turoctocog alfa pegol with other EHL-rFVIII products was available, so PK parameters for turoctocog alfa pegol were assumed equivalent to those of damoctocog alfa pegol, because the two products are the only that have showed the possibility to be administrered once a week. Cl and Vss parameters for the products are reported in Table I.
|
Comparator |
Clearance (mL/h/kg) |
Vss (mL/kg) |
Source |
||
|
Mean |
SD |
Mean |
SD |
||
|
Turoctocog alfa pegol |
1.658 |
0.206 |
40.698 |
0.605 |
Assumption: equal to damoctocog alfa pegol |
|
Rurioctocog alfa pegol |
2.044 |
0.218 |
45.021 |
0.629 |
[15]–recalibrated |
|
Efmoroctocog alfa |
2.073 |
0.198 |
45.215 |
0.774 |
[14]–recalibrated |
|
Damoctocog alfa pegol |
1.658 |
0.206 |
40.698 |
0.605 |
[15]–recalibrated |
Table I. Pharmacokinetics model input parameters for Extending Half-Life (EHL) FVIII products
SD: standard error; Vss: volume of distribution at steady state
IU consumptions and cost estimation (second level)
The second level of the model compares the three setups defined by trough levels of 1%, 3%, and 5% using 1% as reference. Specifically, it computes the annual number of IUs of EHL-rFVIII products and related costs to sustain the defined trough level at the above-described dose frequency for a patient with a mean weight of 74.1 kg as performed in previous economic evaluation of prophylaxis in Italy [16]. The unit costs for EHL-rFVIII products imputed in the model were extracted from So.re.sa (Società Regionale per la Sanità - https://siaps.soresa.it/portalegare/index.php/bandi) and refer to August 2024 prices.
The model also computes the annual treatment cost in Italy for each setup (trough level of 1%, 3% and 5%) simulating the Italian market share: 20% turoctocog alfa pegol, 10% rurioctocog alfa pegol, 50% efmoroctocog alfa, and 20% damoctocog alfa pegol. The difference in annual cost (∆costs) was computed using the 1% trough level as reference. Costs have been computed on a per-patient basis.
A One-Way Sensitivity Analysis (OWSA) was applied to evaluate the impact on ∆costs of varying the value of one model parameter at a time, while holding all other parameters constant [17]. The range of variation of each parameter was used to compute the variation on ∆costs. In detail, the lower and upper values of change on ∆costs were estimated based on the mean value and the relative standard error (SE). The resulting variations of the ∆costs were ranked by their absolute magnitude and were presented in a tornado diagram, highlighting the 17 most influential parameters.
Moreover, an alternative scenario analysis was conducted increasing dose frequencies of EHL-rFVIII products as follows: twice a week for turoctocog alfa pegol, rurioctocog alfa pegol and damoctocog alfa pegol, and every 3 days for efmoroctocog alfa.
Results
The dose required to achieve a specified FVIII level increased as the target trough level increased (Figure 1). For turoctocog alfa pegol (administrered every four days) and rurioctocog alfa pegol (twice weekly), the estimated doses were 20 IU/kg to sustain a 1% trough level, and 60 and 100 IU/kg for 3% and 5% trough levels, respectively. The dose required to maintain the 5% trough level exceeded the maximum dose indicated in the SmPC. For efmoroctocog alfa, the estimated doses were 26, 77 and 128 IU/kg administered 1.9 times per week, with doses greater than 65 IU/kg exceeding the maximum dose indicated in the SmPC. The dose of damoctocog alfa pegol (infusion every 5 days) was 54 IU/kg for achieving 1% trough level and reached 161 and 268 IU/kg for sustaining 3% and 5% trough levels, both over the maximum dose indicated in the SmPC.
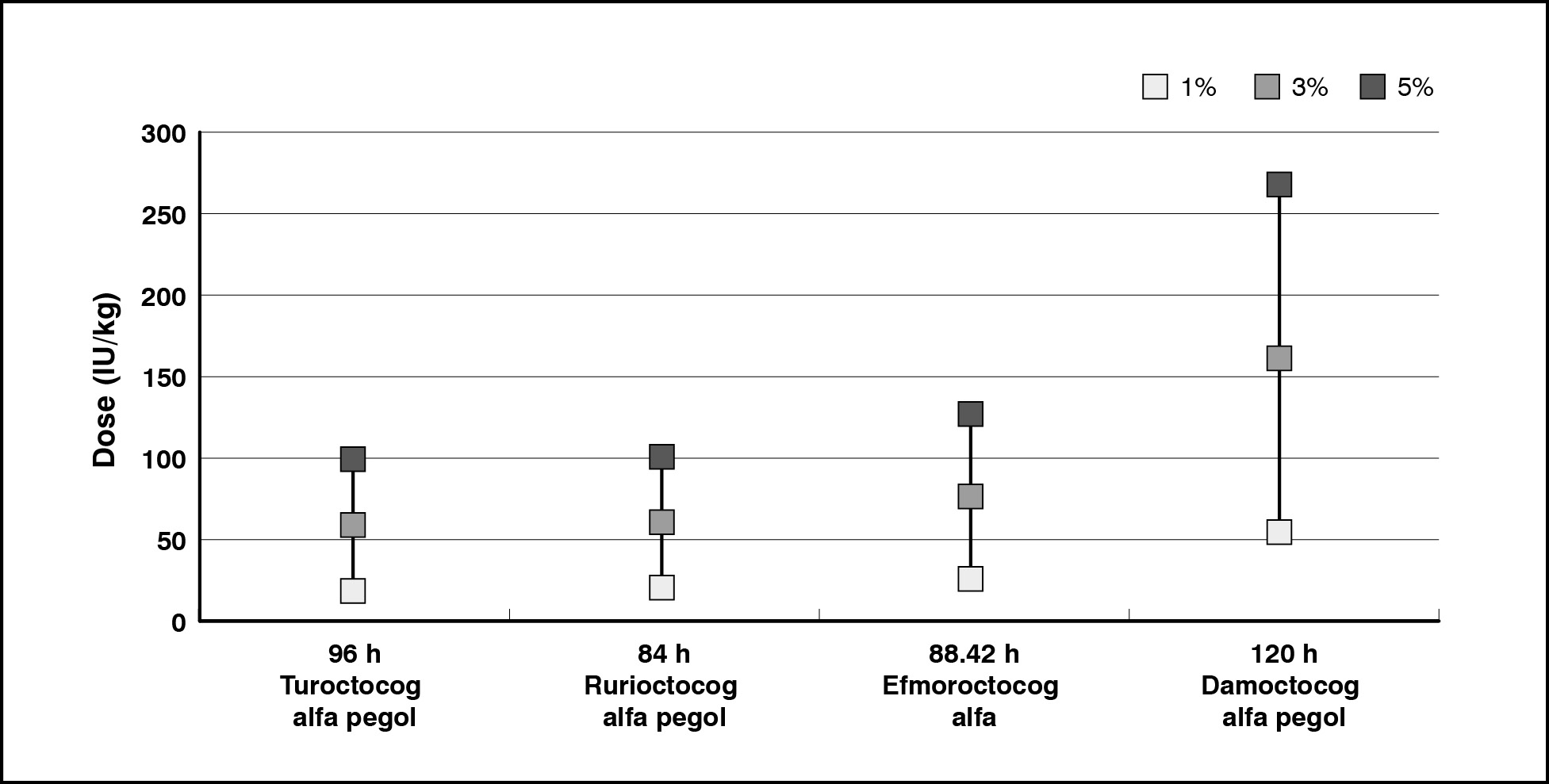
Figure 1. EHL FVIII products dose required to maintain a trough level of 1%, 3%, 5%
The relative annual per-patient treatment cost increased as the trough level increased (Table II). In detail, the per-patient annual cost (for a patient of 74.1 kg) was €117,811 to achieve a trough level of 1%, €351,543 to achieve a 3% trough level and €585,905 to achieve a 5% trough level in the Italian context. That is an increase of 200% (€234,362) of the per-patient cost for sustaining a 3% FVIII level and 400% (€468,724) for a 5% FVIII level. The relative annual per-patient number of IUs for treatment of a patient with mean weight of 74.1kg is reported in Supplementary Table S1.
|
EHL-FVIII product |
Dose frequency (hours) |
Trough level |
Trough level |
|||
|
1% |
3% |
5% |
3% |
5% |
||
|
Annual cost per-patient (€) |
Cost difference vs trough level 1% (€) |
|||||
|
Turoctocog alfa pegol |
96.0 |
63,073 |
189,220 |
315,367 |
126,147 |
252,293 |
|
Rurioctocog alfa pegol |
84.0 |
86,614 |
259,842 |
433,069 |
173,228 |
346,455 |
|
Efmoroctocog alfa |
88.4 |
122,146 |
366,438 |
610,730 |
244,292 |
488,584 |
|
Damoctocog alfa pegol |
120.0 |
174,160 |
522,479 |
870,799 |
348,320 |
696,639 |
|
Market Share |
- |
117,811 |
351,543 |
585,905 |
234,362 |
468,724 |
Table II. Annual cost per-patient of EHL FVIII products to sustain different trough levels
The OWSA on ∆costs revealed that Cl and patient’s body weight have the main impact on cost variations, followed by treatments cost per IU and the market share (Supplementary Figure S2).
The increase in dosing frequency (alternative scenario), that is the reduction of time between infusions, reduced the estimated doses to achieve the pre-defined FVIII levels (Figure 2). The estimated doses remained ≤60 IU/kg for products with increased dose frequency. Therefore, per-patient annual cost decreased to €63,205 when the aim was a 1% trough level, and €189,614 and €316,024 for 3% and 5% trough levels, respectively. This resulted in a ∆costs of €126,410 to shift from a 1% trough level to a 3%, and €252,819 to shift from 1% to 5% (Table III).
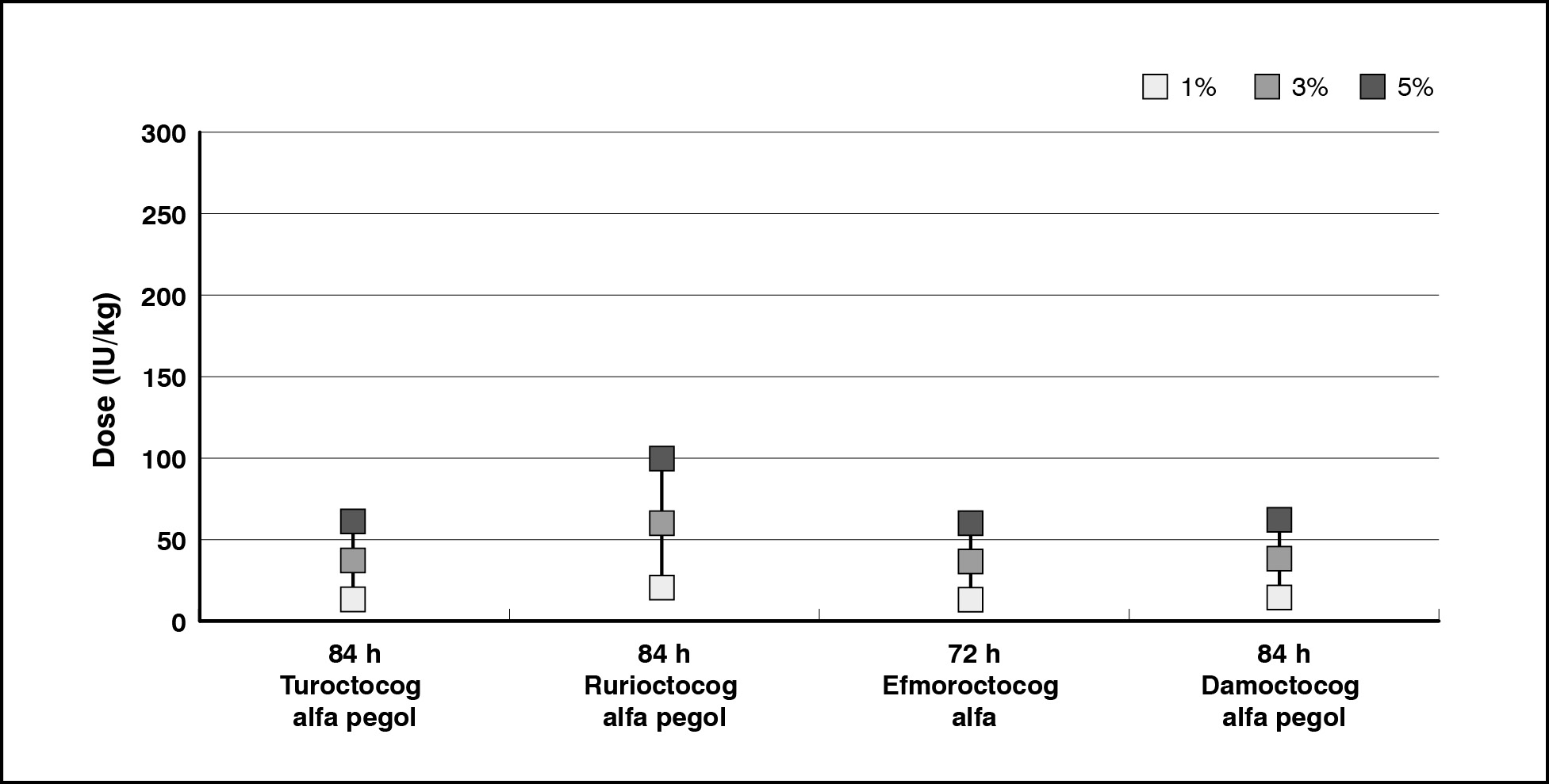
Figure 2. EHL FVIII products dose required to maintain a trough level of 1%, 3%, 5% (alternative scenario)
|
EHL FVIII product |
Dose frequency (hours) |
Trough level |
Trough level |
|||
|
1% |
3% |
5% |
3% |
5% |
||
|
Annual cost per-patient (€) |
Cost difference vs trough level 1% (€) |
|||||
|
Turoctocog alfa pegol |
84.0 |
43,638 |
130,914 |
218,190 |
87,276 |
174,552 |
|
Rurioctocog alfa pegol |
84.0 |
86,614 |
259,842 |
433,069 |
173,228 |
346,455 |
|
Efmoroctocog alfa |
72.0 |
69,257 |
207,772 |
346,286 |
138,514 |
277,029 |
|
Damoctocog alfa pegol |
84.0 |
55,936 |
167,808 |
279,680 |
111,872 |
223,744 |
|
Market Share |
- |
63,205 |
189,614 |
316,024 |
126,410 |
252,819 |
Table III. Annual per-patient cost of EHL FVIII products to sustain different trough levels (alternative scenario)
Discussion
The pharmacokinetics characteristics of SHL-rFVIII and plasma-derived FVIII products and the associated treatment burden (number of intravenous administrations per week), have limited the possibility of achieving trough plasma levels higher than 1-2% and, consequently, higher efficacy [6,7]. New prophylaxis approaches proposed by the World Federation of Hemophilia (WFH) guidelines and expert consensus, based on available EHL-rFVIII, suggest possible new plasma FVIII trough levels targets of at least 3-5% [4,7], with the potential to increase the protection of patients against bleeding, preserving joint status and improving their daily activities. However, reaching these new targets requires an increase in IU consumption and/or administration frequencies, potentially offsetting the possible advantage of treatment burden reduction.
Our study shows how moving from a target of 1% to a target of 3% or 5% could require an increase in IU consumption that is particularly higher if the target is 5%. The IU doses estimated are several times over the maximum dose indicated in the SmPC, are not feasible and are associated with an increased prophylaxis cost of two-to-four times. A possibility to mitigate these increments and use a feasible dose could be an increase in administration frequency. However, even in this scenario, an average cost of €316,024 is required to achieve a trough level of 5%. These results suggest the need for further discussion about the trough levels to target with the current EHL-rFVIII and the prophylaxis dose and regimen selection for each patient, also considering the possibility of using alternative treatment as non-replacement therapy and gene therapy. Furthermore, the theoretical IU consumption estimated by this model should be compared with real-world data to better understand the practical limitations of the new trough level targets recommended by recent guidelines and expert consensus.
In the literature, several real-world studies have assessed the IU consumption and costs associated with the EHL-rFVIII [18-21]; however, these studies have focused mainly on assessing the differences in IU consumptions, costs, and clinical outcomes in patients switching from SHL-rFVIII to EHL-rFVIII products. A study conducted on two U.S. claims databases (the Optum Clinformatics Data Mart and Truven Health MarketScan Databases) analyzed factor IUs dispensed and expenditures for patients with data for ≥3 months before and after switching to an EHL product. Twenty-nine Truven patients switched to an EHL product reporting variable factor IUs dispensed and consistently higher expenditures [18]. In a single-center retrospective study, rFVIII-Fc prophylaxis outcomes were compared with those from the last 18-month SHL-rFVIII prophylaxis. Patients treated with rFVIII-Fc prophylaxis reported comparable FVIII weekly dose and reduced infusion frequency (mean −30%) compared with previous SHL-rFVIII prophylaxis thrice weekly or every other day. In 13 patients, over a mean 18-month follow-up, significantly lower FVIII weekly dose and annual consumption (mean −12%; p=0.019), comparable bleeding rates and FVIII trough levels, and improved management of breakthrough bleeding were observed [19]. A retrospective inception cohort of all adult hemophilia A patients switched to EHL-rFVIII (rFVIIIFc or PEGylated rFVIII) prophylaxis was conducted in one center in Finland to assess the differences in dosing, product utilization, annualized bleed rates (ABR), treatment regimen and pharmacokinetics between SHL-rFVIII and EHL-rFVIII. The study reported a median FVIII dose of 23 IU/kg for SHL versus 25 IU/kg for EHL and a weekly infusion decreas of 29% from median 2.8 (every 2.5 days) to 2.0 (every 3.5 days) (p≤0.001). Overall, median weekly FVIII consumption decreased after switching by 17% from 60 to 50 IU/kg (p=0.001): weekly product utilization decreased by at least 5 IU/kg in 55%, remained the same in 32% but increased by at least 5 IU/kg in 13%. The half-life of FVIII increased from a median of 13 to 21 h after switching, as did the times above 1% and 3 % FVIII threshold, which improved from 85 to 131 h and from 65 to 106 h [20]. Another study conducted in 7 centers in Austria compared clinical outcomes and factor utilization in patients with SHA, who switched from prophylaxis with SHL-rFVIII to an EHL-rFVIII. Twenty patients reported a significantly decreased annualized bleeding rate [ABR; median difference (IQR): −0.3 (−4.5; –0); p=0.008] and number of prophylactic infusions per week [−0.75 (−1.0; –0.0); p=0.007] after switching to rFVIII-Fc. While factor utilization was comparable to prior prophylaxis with SHL [0.0 (−15.8; –24.8) IU/kg/week; p=0.691] [21]. All these studies showed variability in outcomes and costs associated with the switch from SHL-rFVIII to EHL-rFVIII. The average results suggest a possible improvement in clinical outcomes with no change or an increase in IU consumption. Unfortunately, these studies do not specifically assess the impact of targeting a 3% or 5% trough level, as done in our analysis, and cannot be directly compared with our results. However, these real-world data indicate a possible use of EHL-rFVIII to increase the efficacy without reducing the IU consumption. Furthermore, real-world data reflect the individualized approach adopted by hemophilia centers and clinicians in defining optimal treatment regimens, in contrast to the hypothetical scenarios presented in model-based analyses. These two approaches serve different purposes and should be interpreted according to their respective study designs: while a model-based analysis is designed to address the lack of real-world evidence and evaluate scenarios that are difficult or impossible to test empirically, real-world studies are conducted to validate model predictions and assess how factors excluded from theoretical models influence outcomes.
Finally, we are aware of several limitations of our study. First, the pharmacokinetics parameters can vary significantly between patients and the average results produced by this study are not applicable to the individual patient with hemophilia. However, even if the results of our study could overestimate or underestimate the real IU consumption required for the single patient, potentially creating clinically unrealistic dosing scenarios, the overall impact on the population will tend toward the average, resulting in estimates more in line with our findings. Indeed, the use of new EHL-rFVIII appears to be leaning towards higher protection against bleedings instead of reducing the treatment burden and IU consumption. This statement is supported by the data of a recent report on Italian FVIII consumption, which reported a limited reduction in the IU of FVIII used after the introduction of different EHL-rFVIII products [22]. Second, the analysis is based on a theoretical PK model that includes certain assumptions (e.g., PK equivalence between turoctocog alfa pegol and damoctocog alfa pegol, and the use of average PK parameters instead of individual PK characteristics). These assumptions limit the possibility of including inter-patient variability and product-specific differences that would allow for a more accurate, patient-specific assessment. To overcome these limitations, real-world studies are required to assess the impact of EHL-rFVIII on individual IU consumption and FVIII trough levels achieved. However, real-world data regarding the changes in trough levels achieved in Italian patients and the associated consumption of rFVIII IU are lacking; this study represent a first attempt to fill this gap.
In conclusion, our results demonstrate a significant increase in mean IU consumption and associated costs when the trough level target is set to 3% or 5% for the hemophilia A population, according to our theoretical PK model. The required IU doses often exceed SmPC maximum doses, rendering these regimens clinically infeasible. While increasing administration frequency can mitigate these issues, the cost per-patient remains significantly high (e.g. an average cost of €316,024 per-patient-year for a 5% trough level). These findings emphasize the importance of individualizing clinical and economic feasibility assessments for new trough targets, ensuring that guidelines are adapted to each patient’s unique profile and therapeutic goals. Furthermore, careful consideration should be given to alternative treatment options (e.g., non replacement factors, gene therapy), to identify the most cost-effective and beneficial approach for each individual. Real-world studies are necessary to confirm our findings and to accurately measure the real-world impact of EHL-rFVIII products in achieving higher levels of protection.
Author Contributions
Study conception and design: C.F., L.G.M., G.C., P.A.C. Model development: C.F., L.G.M., P.A.C. Model validation: S.A., S.S., G.C. Draft manuscript preparation: C.F., P.A.C.. All authors critically revised the manuscript, reviewed the results, and approved the final version of the manuscript.
Funding
The design of the model that support the findings was funded by Roche Italy S.p.A
Conflict of interest
CF, SA and SS have no conflict of interest. GC p received payment or honoraria for lectures, presentations, speakers’ bureau, or educational events Bayer, Bioviiix, CSL Behring, Biomarin, Sanofi, Novo Nordisk, Takeda, LFB, Roche and SOBI; participation on a Data Safety Monitoring Board or Advisory Board Bayer, CSL Behring, Biomarin, Sanofi, Novo Nordisk, Takeda, LFB and Roche. PAC has received honoraria for lectures, presentations, speakers’ bureau, or educational events from Roche, Novartis, Bayer, Almirall, Daiichi-Sankyo and Otsuka. LGM has received grants and personal fees from Bayer AG, Boehringer Ingelheim, Pfizer and Daiichi-Sankyo.
References
1.Mannucci PM, Tuddenham EG. The hemophilias--from royal genes to gene therapy. N Engl J Med. 2001;344(23):1773-1779; https://doi.org/10.1056/NEJM200106073442307
2.Manco-Johnson MJ, Abshire TC, Shapiro AD, et al. Prophylaxis versus episodic treatment to prevent joint disease in boys with severe hemophilia. N Engl J Med. 2007;357(6):535-544; https://doi.org/10.1056/NEJMoa067659
3.Andersson NG, Auerswald G, Barnes C, et al. Intracranial haemorrhage in children and adolescents with severe haemophilia A or B - the impact of prophylactic treatment. Br J Haematol. 2017;179(2):298-307; https://doi.org/10.1111/bjh.14844
4.Srivastava A, Santagostino E, Dougall A, et al. WFH Guidelines for the Management of Hemophilia, 3rd edition. Haemophilia. 2020;26 Suppl 6:1-158; https://doi.org/10.1111/hae.14046
5.Berntorp E, Andersson NG. Prophylaxis for Hemophilia in the Era of Extended Half-Life Factor VIII/Factor IX Products. Semin Thromb Hemost. 2016;42(5):518-525; https://doi.org/10.1055/s-0036-1571315
6.Collins PW, Blanchette VS, Fischer K, et al. Break-through bleeding in relation to predicted factor VIII levels in patients receiving prophylactic treatment for severe hemophilia A. J Thromb Haemost. 2009;7(3):413-420; https://doi.org/10.1111/j.1538-7836.2008.03270.x
7.Peyvandi F, Berger K, Seitz R, et al. Kreuth V initiative: European consensus proposals for treatment of hemophilia using standard products, extended half-life coagulation factor concentrates and non-replacement therapies. Haematologica. 2020;105(8):2038-2043; https://doi.org/10.3324/haematol.2019.242735
8.Klamroth R, Windyga J, Radulescu V, et al. rurioctocog alfa pegol PK-guided prophylaxis in hemophilia A: results from the phase 3 PROPEL study. Blood. 2021;137(13):1818-1827; https://doi.org/10.1182/blood.2020005673 rurioctocog
9.Benson G, Morton T, Thomas H, Lee XY. Long-Term Outcomes of Previously Treated Adult and Adolescent Patients with Severe Hemophilia A Receiving Prophylaxis with Extended Half-Life FVIII Treatments: An Economic Analysis from a United Kingdom Perspective. Clinicoecon Outcomes Res. 2021;13:39-51. https://doi.org/10.2147/CEOR.S280574
10.Mahlangu J, Powell JS, Ragni MV, et al.; A-LONG Investigators. Phase 3 study of recombinant factor VIII Fc fusion protein in severe hemophilia A. Blood. 2014;123(3):317-325; https://doi.org/10.1182/blood-2013-10-529974
11.SmPC Turoctocog alfa pegol - Esperoct. Available at: https://farmaci.agenziafarmaco.gov.it/aifa/servlet/PdfDownloadServlet?pdfFileName=footer_000085_048083_RCP.pdf&sys=m0b1l3
12.SmPC Rurioctocog alfa pegol - Adynovate. Available at: https://farmaci.agenziafarmaco.gov.it/aifa/servlet/PdfDownloadServlet?pdfFileName=footer_003822_045936_RCP.pdf&sys=m0b1l3
13.SmPC Damoctocog alfa pegol - Jivi. Available at: https://farmaci.agenziafarmaco.gov.it/aifa/servlet/PdfDownloadServlet?pdfFileName=footer_000689_047418_FI.pdf&sys=m0b1l3
14.Shah A, Solms A, Wiegmann S, et al. Direct comparison of two extended-half-life recombinant FVIII products: a randomized, crossover pharmacokinetic study in patients with severe hemophilia A. Ann Hematol. 2019;98(9):2035-2044; https://doi.org/10.1007/s00277-019-03747-2
15.Solms A, Shah A, Berntorp E, et al. Direct comparison of two extended half-life PEGylated recombinant FVIII products: a randomized, crossover pharmacokinetic study in patients with severe hemophilia A. Ann Hematol. 2020;99(11):2689-2698; https://doi.org/10.1007/s00277-020-04280-3
16.Cortesi PA, Castaman G, Trifirò G, et al. Cost-Effectiveness and Budget Impact of Emicizumab Prophylaxis in Haemophilia A Patients with Inhibitors. Thromb Haemost. 2020;120(2):216-228; https://doi.org/10.1055/s-0039-3401822
17.Drummond MF, Sculpher MJ, Claxton K, et al. Methods for the Economic Evaluation of Health Care Programmes. Oxford: Oxford University Press, 2015
18.Chhabra A, Fogarty PF, Tortella BJ, et al. Real-World Analysis of Dispensed International Units of Coagulation Factor VIII and Resultant Expenditures for Hemophilia A Patients: A Comparison Between Standard Half-Life and Extended Half-Life Products. Manag Care. 2018;27(10):39-50.
19.Tagliaferri A, Matichecchia A, Rivolta GF, et al. Optimising prophylaxis outcomes and costs in haemophilia patients switching to recombinant FVIII-Fc: a single-centre real-world experience. Blood Transfus. 2020;18(5):374-385; https://doi.org/10.2450/2019.0220-19
20.Nummi V, Lehtinen AE, Iorio A, et al. Switching from standard to extended half-life FVIII prophylaxis in haemophilia A: Comparison of factor product use, bleed rates and pharmacokinetics. Haemophilia. 2022;28(6):e237-e244; https://doi.org/10.1111/hae.14649; https://doi.org/
21.Ay C, Feistritzer C, Rettl J, et al. Bleeding outcomes and factor utilization after switching to an extended half-life product for prophylaxis in haemophilia A in Austria. Sci Rep. 2021;11(1):12967; https://doi.org/10.1038/s41598-021-92245-5
22.Candura F, Massari MS, Profili S, et al. Analisi della domanda di medicinali plasmaderivati in Italia. 2022. Roma: Istituto Superiore di Sanità; 2023. (Rapporti ISTISAN 23/31 IT)